
CSD complex facilities (sterilization equipment)
CSD complex facilities (sterilization equipment)
At present, hospital-acquired infections (HAI) are one of the most pressing problems of medicine. According to official statistics, 50,000 - 60,000 cases of HAI occur annually in Russia, but in reality the number of patients infected with this infection is close to 3 million people. At the same time, there is an annual increase in the number of cases of HAI by 1%. Among medical workers, the HAI is even more common.
Correct organization of the sterilization process is the best way to fight hospital-acquired infections. In turn, the effectiveness of the measures carried out depends on the availability of appropriate technical capabilities regarding the mechanization and automation of the sterilization process. It is believed that in almost 50% of cases, this infection is transmitted parenterally, with the medical instrumentation playing a leading role. For this reason, a properly organized sterilization department is the "reliable rear" of any hospital in a battle with a hospital-acquired infections, and the complex equipment of the Sterile Services Department plays an important role.
What we do
Pharmstandard-Medtechnika offers its services in the reconstruction and building of new sterilization departments, as well as carries out complex equipment of CSD, as requested by the sterilization departments of healthcare facilities in accordance with GOSTs. The central sterilization departments are equipped with everything necessary for the continuous processing of the sterilized material and tools, including furniture, sorting and packing tables, thermal sealing machines, ultrasonic washing machines, drying modules, washing and disinfection machines, sterilization baskets, shelves and cabinets for storage of sterile materials, containers for transportation of sterile materials, and pass-through hatches. for the continuous processing of the sterilized material and tools, including furniture, sorting and packing tables, thermal sealing machines, ultrasonic washing machines, drying modules, washing and disinfection machines, sterilization baskets, shelves and cabinets for storage of sterile materials, containers for transportation of sterile materials, and pass-through hatches.
Currently, several hundred CSDs have been created in Russia using the sterilization equipment supplied by Pharmstandard-Medtechnika LLC. All activities are tailored to the specific needs of the healthcare institution.
Our advantages. What does the healthcare institution get?
- State-of-the-art equipment at a low cost (as compared to other foreign suppliers).
- Accomplishing all the tasks set by the customer in a fast and effective way at any stage of the project.
- Significant advantages in cost, timing and quality of the project due to the fact that Pharmstandard-Medtechnika is responsible for the whole project.
- Productive innovative equipment that is easy and simple to operate as it is designed taking into account specific Russian requirements and the design works, logistics and maintenance are completely localized.
- Confidence in a high quality support of equipment operation. The Technical Department of Pharmstandard-Medtechnika LLC performs all the necessary works, provides consultations and promptly responds to customer requests.
- Uninterrupted operation of equipment and quick delivery of spare parts when necessary.
- High economic effect based on reliable equipment operation, low power consumption and minimal maintenance costs.
According to the regulatory documents (MR No 15-6/8 dated 01.02.90 "Methodological recommendations for the organization of centralized sterilization in healthcare facilities", "Manual for the design of healthcare facilities (to SniP2.08. 02-89 Section 3 "Specialized, auxiliary subdivisions and utility rooms"), MU 287-113-98 "Methodological guidelines for disinfection, pre-sterilization cleaning and sterilization of medical devices", GOST R 51935-2002 (EN 285) "Steam large sterilizers. General technical requirements and test methods”, GOST R ISO 13683–2000 “Sterilization of health care products.” Requirements for validation and routine control of moist heat sterilization in health care facilities.”), modern CSD should meet the following requirements:
Modern Requirements To The Csd
The basis of CSD is modern sterilization equipment, which must meet the following requirements:
- Automatic microprocessor control
- Forced air removal from the sterilization chamber (pre-vacuum)
- Through-type sterilization chamber
- Regimes approved by the Ministry of Health of Russian Federation
- Freely programmable and test regimes
- Availability of devices for documenting and recording sterilization process parameters
- Safety system
- Sterilization chamber and the main components must be made of at least stainless steel 08X18H10 (AISI 304); for sterilization in corrosive environments - at least 03X17H14M2 (AISI 316L)
- Sterilizers must meet the requirements of GOST R 51935–2002 (EN 285)
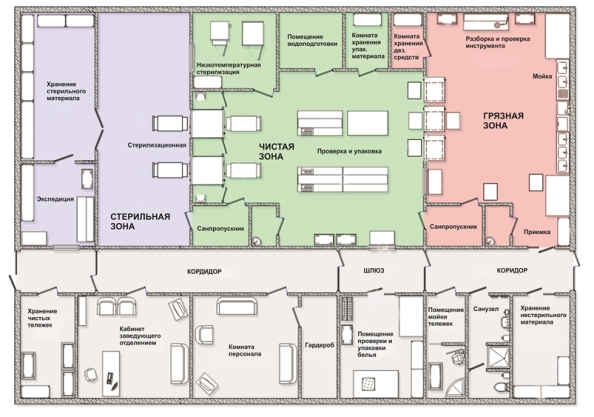
In a modern CSD there should be a clear division of production areas into "dirty", "clean" and "sterile" zones.
When restrictions exist, it is allowed to divide into "non-sterile" and "sterile" zones. The traffic flows do not intersect, which eliminates the risk of re-contamination of sterile medical devices. The barrier between the "dirty" and "clean" zones is the through-type washing and disinfection plants; between the "clean" and "sterile" zones - through-type sterilization equipment. Functional connection between zones is carried out through a special gateway.All technological equipment used in the CSD should have automatic control, as well as the function of documenting sterilization parameters to ensure postoperative quality control of processed products.
In combination with the automation of the main processes, this ensures quality pre-sterilization cleaning and sterilization.Pre-sterilization cleaning of medical devices should be carried out using automatic through-type washing and disinfection equipment.
These processes must be provided with distilled water to ensure quality pre-sterilization and sterilization in the CSD. It is recommended to use distilled water, because ordinary tap water contains salts (chlorides), which cause corrosion of the treated metal tools and process equipment.
Stages of csd creation
-
Design development
The design development stage starts with collection of information and materials used in preparation of the technical part of the project. If necessary, specialists will visit the facility. Proposals on process layout, room plans, and design assignment on engineering communications are being developed. Modern requirements to CSD should be taken into account during these works. Planning solutions and regimes of technological processes of products and materials processing are developed taking into account the requirements of regulatory documentation.
-
Comprehensive fit out
The comprehensive fit out is the supply of main technological and auxiliary equipment, including transport facilities, engineering facilities, and production furniture.State of the art steam sterilization equipment offered by Pharmstandard-Medtechnika fully complies with the requirements of GOST R 51935-2002 (EN 285).
Submit a request for our services:
Or contact us:
Phone
8 (495) 739-39-45




